Sea Surface Temperature - Real-time
Details
Permalink to Details- Added to the Catalog
- Available for
- SOS
- Explorer
- Categories
- Water: Temperature
- Keywords
- Oceans
- Real-time
- Sea Surface Temperature
- Seasons
- SST
- Temperature
Description
Permalink to DescriptionSea surface temperature, much like the atmosphere’s temperature, is constantly changing. Water warms up and cools down at a slower rate than air, so diurnal variations (heating during the day and cooling during the night) seen in the atmosphere are hard to observe in the ocean. The seasons, however, can be seen as the warmest water near the equator expands toward the United States during the summer months and withdraws again during the winter months. The interaction between the ocean and the atmosphere is one that scientists are constantly researching, and the temperature of the sea surface is a key factor in those interactions. Sea surface temperature anomaly is the difference between the current temperature and the long-term temperature average. Negative temperature differences indicate that the ocean is cooler than average, while positive temperature difference indicate that the ocean is warmer than average. Tracking sea surface anomalies helps scientists quickly identify areas of warming and cooling, which can effect coral reef ecosystems, hurricane development, and the development of El Nino and La Nina.
The real-time sea surface temperature dataset is provided by the NOAA Visualization Lab using data from NOAA’s Coral Reef Watch. These highly detailed, daily images are possible because the data combines SST measurements from all current geostationary and polar-orbiting satellites, including U.S. satellites and those of international partners such as Japan and Europe. By collecting data from multiple satellites, cloud-free composites can be generated on a daily basis because at some point, one of the satellites will get a cloud-free observation of the ocean surface during the 24-hour period. The resolution of the data is 5 km/pixel. At this resolution, features such as the Gulf Stream along the East Coast of the United States are distinguishable.
The entire archive of this imagery, starting in 1981, is available from NOAA View or FTP for download to your SOS computer.
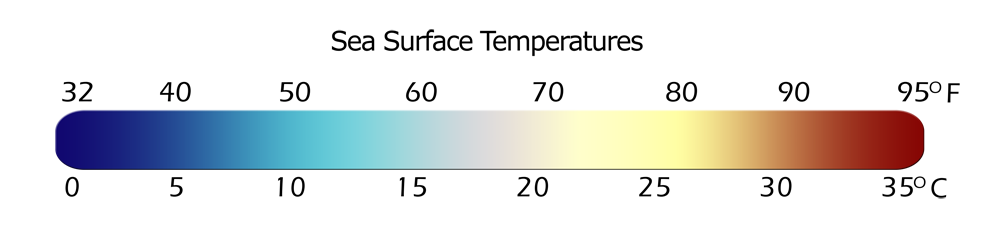
Next Generation Science Standards
Permalink to Next Generation Science StandardsCross-cutting Concepts
Permalink to Cross-cutting ConceptsGrades 3–5
C1 Patterns. Students identify similarities and differences in order to sort and classify natural objects and designed products. They identify patterns related to time, including simple rates of change and cycles, and to use these patterns to make predictions.
C2 Cause and Effect. Students routinely identify and test causal relationships and use these relationships to explain change. They understand events that occur together with regularity might or might not signify a cause and effect relationship
C3 Scale Proportion and Quantity. Students recognize natural objects and observable phenomena exist from the very small to the immensely large. They use standard units to measure and describe physical quantities such as weight, time, temperature, and volume.
C5 Energy and Matter. Students learn matter is made of particles and energy can be transferred in various ways and between objects. Students observe the conservation of matter by tracking matter flows and cycles before and after processes and recognizing the total weight of substances does not change.
Grades 6–8
C1 Patterns. Students recognize that macroscopic patterns are related to the nature of microscopic and atomic-level structure. They identify patterns in rates of change and other numerical relationships that provide information about natural and human designed systems. They use patterns to identify cause and effect relationships, and use graphs and charts to identify patterns in data.
C2 Cause and Effect. Students classify relationships as causal or correlational, and recognize that correlation does not necessarily imply causation. They use cause and effect relationships to predict phenomena in natural or designed systems. They also understand that phenomena may have more than one cause, and some cause and effect relationships in systems can only be described using probability.
C3 Scale Proportion and Quantity. Students observe time, space, and energy phenomena at various scales using models to study systems that are too large or too small. They understand phenomena observed at one scale may not be observable at another scale, and the function of natural and designed systems may change with scale. They use proportional relationships (e.g., speed as the ratio of distance traveled to time taken) to gather information about the magnitude of properties and processes. They represent scientific relationships through the use of algebraic expressions and equations
C5 Energy and Matter. Students learn matter is conserved because atoms are conserved in physical and chemical processes. They also learn within a natural or designed system, the transfer of energy drives the motion and/or cycling of matter. Energy may take different forms (e.g. energy in fields, thermal energy, energy of motion). The transfer of energy can be tracked as energy flows through a designed or natural system.
Grades 9–12
C1 Patterns. Students observe patterns in systems at different scales and cite patterns as empirical evidence for causality in supporting their explanations of phenomena. They recognize classifications or explanations used at one scale may not be useful or need revision using a different scale; thus requiring improved investigations and experiments. They use mathematical representations to identify certain patterns and analyze patterns of performance in order to re-engineer and improve a designed system.
C5 Energy and Matter. Students learn that the total amount of energy and matter in closed systems is conserved. They can describe changes of energy and matter in a system in terms of energy and matter flows into, out of, and within that system. They also learn that energy cannot be created or destroyed. It only moves between one place and another place, between objects and/or fields, or between systems. Energy drives the cycling of matter within and between systems. In nuclear processes, atoms are not conserved, but the total number of protons plus neutrons is conserved.
C7 Stability and Change. Students understand much of science deals with constructing explanations of how things change and how they remain stable. They quantify and model changes in systems over very short or very long periods of time. They see some changes are irreversible, and negative feedback can stabilize a system, while positive feedback can destabilize it. They recognize systems can be designed for greater or lesser stability
Disciplinary Core Ideas
Permalink to Disciplinary Core IdeasGrades 3–5
ESS2.A Earth Materials and Systems. Four major Earth systems interact. Rainfall helps to shape the land and affects the types of living things found in a region. Water, ice, wind, organisms, and gravity break rocks, soils, and sediments into smaller pieces and move them around
ESS2.C The Roles of Water in Earth's Processes. Most of Earth’s water is in the ocean and much of the Earth’s fresh water is in glaciers or underground.
ESS2.D Weather & Climate. Climate describes patterns of typical weather conditions over different scales and variations. Historical weather patterns can be analyzed so that they can make predictions about what kind of weather might happen next.
Grades 6–8
ESS2.A Earth Materials and Systems. Energy flows and matter cycles within and among Earth’s systems, including the sun and Earth’s interior as primary energy sources. Plate tectonics is one result of these processes.
ESS2.C The Roles of Water in Earth's Processes. Water cycles among land, ocean, and atmosphere, and is propelled by sunlight and gravity. Density variations of sea water drive interconnected ocean currents. Water movement causes weathering and erosion, changing landscape features.
ESS2.D Weather & Climate. Complex interactions determine local weather patterns and influence climate, including the role of the ocean.
ESS3.D Global Climate Change. Human activities affect global warming. Decisions to reduce the impact of global warming depend on understanding climate science, engineering capabilities, and social dynamics.
PS3.A Definitions of Energy. Kinetic energy can be distinguished from the various forms of potential energy. Energy changes to and from each type can be tracked through physical or chemical interactions. The relationship between the temperature and the total energy of a system depends on the types, states, and amounts of matter.
Grades 9–12
ESS2.A Earth Materials and Systems. Feedback effects exist within and among Earth’s systems.The geological record shows that changes to global and regional climate can be caused by interactions among changes in the sun’s energy output or Earth’s orbit, tectonic events, ocean circulation, volcanic activity, glaciers, vegetation, and human activities.
ESS2.C The Roles of Water in Earth's Processes. The planet’s dynamics are greatly influenced by water’s unique chemical and physical properties.
ESS2.D Weather & Climate. The role of radiation from the sun and its interactions with the atmosphere, ocean, and land are the foundation for the global climate system. Global climate models are used to predict future changes, including changes influenced by human behavior and natural factors
ESS3.D Global Climate Change. Global climate models used to predict changes continue to be improved, although discoveries about the global climate system are ongoing and continually needed.
PS3.A Definitions of Energy. The total energy within a system is conserved. Energy transfer within and between systems can be described and predicted in terms of energy associated with the motion or configuration of particles (objects).
Notable Features
Permalink to Notable Features- This dataset provides Real Time data:
- Range: 1 year
- Period: 1 day
- Growth and reduction of the warm water during the changing seasons
- Sea surface temperature denoted by colorbar, 35°C = 95°F, 0°C = 32°F