Aurora (3D)
Details
Permalink to Details- Added to the Catalog
- Available for
- Explorer
- Categories
- Space: Earth's Magnetism
- Keywords
- Aurora Australis
- Electrons
- Geomagnetism
- Magnetic Field
- Magnetosphere
Description
Permalink to DescriptionAurora 3D on SOS Explorer:
Permalink to Aurora 3D on SOS Explorer:This graphical volumetric (3D) visualization of the aurora borealis is an artist's rendition and does not reflect real data at any one point in time.
Aurora:
Permalink to Aurora:Galileo first used the term to describe the light. Aurora is the Roman goddess of dawn.
Borealis:
Permalink to Borealis:The Greek name for the north wind (Boreas). In the southern hemisphere it is called Aurora Australis.
What causes the light that we call the aurora:
Permalink to What causes the light that we call the aurora:The light in the upper atmosphere is caused by energetic electrons zipping down the magnetic field lines of Earth and colliding with the atoms and molecules of the atmosphere (primarily O and N2). When the electrons collide with the atmosphere, they transfer momentum energy into excited states of the atom or molecule. When the atoms and molecules relax back to their ground state they release photons that we can see.
Colors of the aurora:
Permalink to Colors of the aurora:The most common color is a pale yellowish green. This color occurs at a very specific wavelength (557.7 nm). It is produced by atomic oxygen. Another common color, is a red glow at 630.0 nm, which is also produced by oxygen. Sometimes the lower edges of aurora rays can be a bluish purple (427.8 nm) from molecular nitrogen. And if you see aurora near sunrise or sunset, the sunlight hits the tops of the auroral rays and ionizes the excited nitrogen to create a purple color. There are many other colors in the aurora but they are either too dim to see with the naked eye or they are outside the visible spectrum, either in the infrared or ultraviolet (where the human eye can't see them).
What causes aurora:
Permalink to What causes aurora:The process of accelerating electrons into the atmosphere starts at the sun. When the sun is active (near solar maximum), it can eject large blobs of plasma called Coronal Mass Ejections (CMEs). A CME is a billion tons of material traveling at up to 2000 km/sec (or 4.5 million miles/hour). If the CME is directed toward Earth, it will take one to three days for it to travel the 150 million km (96 million miles) from sun to Earth. When it reaches Earth, the magnetic field of Earth deflects much of the CME around Earth but some of the more energetic particles can enter the Earth's magnetic field or Magnetosphere. Even though much of the mass is deflected around Earth, the impact can jar the magnetosphere and shake it up, causing it to change configuration. All of this shaking and reconfiguring can accelerate electrons that are normally trapped within the magnetosphere. The accelerated electrons are still confined to follow the magnetic field lines of Earth but now they travel down the field line and collide with the atmosphere. Early scientists made the connection between an increase in sunspots (active regions on the sun) and an increase in the intensity and frequency of the aurora.
During solar minimum, when CMEs are less likely, it is still possible to have a strong aurora. A feature on the sun called a coronal hole is associated with strong background solar wind. Normally the solar wind flows about 300-500 km/sec but from a coronal hole, the solar wind can flow to 800 km/sec. This increase in solar wind speed can also cause the magnetosphere to reconfigure, the electrons to get accelerated, and aurora to form. These auroral displays are less dramatic than those associated with CMEs but they can still be quite spectacular if you are in the right place at the right time.
Geomagnetic Storms:
Permalink to Geomagnetic Storms:When CMEs hit the magnetosphere or when strong solar wind from a coronal hole shakes the magnetosphere, it causes the magnetic field to change and reconfigure. Magnetic field lines can break and reconnect which can in turn accelerate electrons. All of these changes result in a change in the magnetic field of Earth. If you hold a magnetic compass very still during a strong geomagnetic storm, you might observe the needle to move around. One of the consequences of this is that migratory birds that use Earth's magnetic field to know where to fly, will lose their way. Pigeon racers know about this and will not let their pigeons fly during big geomagnetic storms.
Geomagnetic storms are ranked on a scale of 0 to 9 with 9 being the largest. This is called the planetary K index or Kp. During a typical 11 year solar cycle there might be two to four geomagnetic storms that reach Kp = 9.
Where to go to observe the aurora:
Permalink to Where to go to observe the aurora:Because the aurora is trapped on the magnetic field lines, it is most often seen near the north and south poles of Earth. Because of the structure of the magnetosphere, and the primary acceleration processes in the tail part of the magnetosphere (away from the sun), aurora form a ring or oval around each of the poles of Earth and are brightest and most dramatic near midnight. The auroral ovals are not centered on the geographic pole but are centered on the magnetic pole of Earth. On nights where there is little geomagnetic activity and the aurora is quiet, it stays further poleward. But when activity picks up, it becomes brighter and the auroral oval expands towards the equator. During very large events, it is possible to see the aurora in the continental US.
It should be noted that the aurora is symmetric and auroral features in the northern hemisphere are mirrored in the southern hemisphere.
Here are several suggestions on what to consider when looking for aurora. First it must be dark. Traveling to the polar regions in the summer won't work because the sun is up 24 hours a day. Second, it must be clear. Often coastal regions are cloudy, so going someplace away from the oceans improves probabilities of seeing aurora. Third, it is good to get away from other sources of light such as cities, streetlights, and even the full moon. Fourth, you should stay up and watch for aurora between 10 PM and 1 AM local time. That is when the aurora is the brightest and most active. And finally, it is good to have some geomagnetic activity. The aurora is visible somewhere on Earth nearly all the time. But it is usually just a few faint arcs that do not move or change very much. Geomagnetic activity will cause the aurora to get brighter, to move further towards the equator, and to become more active.
You can see the aurora from space. The astronauts on the International Space Station see the aurora all the time and actually fly through the aurora on occasion. There is also aurora on other planets. Jupiter and Saturn have regular auroral displays.
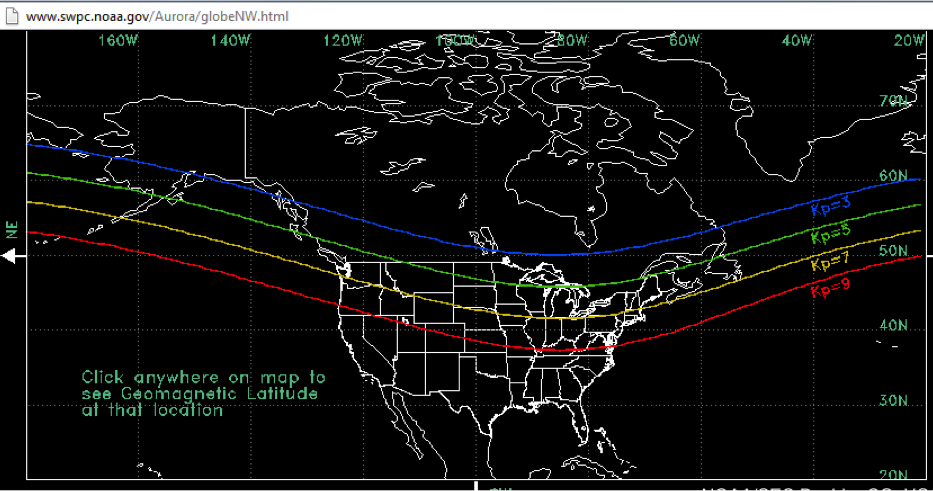
Figure: Where you can see aurora for various levels of geomagnetic activity.
Next Generation Science Standards
Permalink to Next Generation Science StandardsCross-cutting Concepts
Permalink to Cross-cutting ConceptsGrades K–2
C1 Patterns. Children recognize that patterns in the natural and human designed world can be observed, used to describe phenomena, and used as evidence
C3 Scale Proportion and Quantity. Students use relative scales (e.g., bigger and smaller; hotter and colder; faster and slower) to describe objects. They use standard units to measure length.
Grades 3–5
C2 Cause and Effect. Students routinely identify and test causal relationships and use these relationships to explain change. They understand events that occur together with regularity might or might not signify a cause and effect relationship
C5 Energy and Matter. Students learn matter is made of particles and energy can be transferred in various ways and between objects. Students observe the conservation of matter by tracking matter flows and cycles before and after processes and recognizing the total weight of substances does not change.
Grades 6–8
C3 Scale Proportion and Quantity. Students observe time, space, and energy phenomena at various scales using models to study systems that are too large or too small. They understand phenomena observed at one scale may not be observable at another scale, and the function of natural and designed systems may change with scale. They use proportional relationships (e.g., speed as the ratio of distance traveled to time taken) to gather information about the magnitude of properties and processes. They represent scientific relationships through the use of algebraic expressions and equations
C4 Systems and System Models. Students can understand that systems may interact with other systems; they may have sub-systems and be a part of larger complex systems. They can use models to represent systems and their interactions—such as inputs, processes and outputs—and energy, matter, and information flows within systems. They can also learn that models are limited in that they only represent certain aspects of the system under study.
C5 Energy and Matter. Students learn matter is conserved because atoms are conserved in physical and chemical processes. They also learn within a natural or designed system, the transfer of energy drives the motion and/or cycling of matter. Energy may take different forms (e.g. energy in fields, thermal energy, energy of motion). The transfer of energy can be tracked as energy flows through a designed or natural system.
C7 Stability and Change. Students explain stability and change in natural or designed systems by examining changes over time, and considering forces at different scales, including the atomic scale. Students learn changes in one part of a system might cause large changes in another part, systems in dynamic equilibrium are stable due to a balance of feedback mechanisms, and stability might be disturbed by either sudden events or gradual changes that accumulate over time
Grades 9–12
C1 Patterns. Students observe patterns in systems at different scales and cite patterns as empirical evidence for causality in supporting their explanations of phenomena. They recognize classifications or explanations used at one scale may not be useful or need revision using a different scale; thus requiring improved investigations and experiments. They use mathematical representations to identify certain patterns and analyze patterns of performance in order to re-engineer and improve a designed system.
C2 Cause and Effect. Students understand that empirical evidence is required to differentiate between cause and correlation and to make claims about specific causes and effects. They suggest cause and effect relationships to explain and predict behaviors in complex natural and designed systems. They also propose causal relationships by examining what is known about smaller scale mechanisms within the system. They recognize changes in systems may have various causes that may not have equal effects.
Disciplinary Core Ideas
Permalink to Disciplinary Core IdeasGrades K–2
ESS1.B Earth and the Solar System. Patterns of movement of the sun, moon, and stars as seen from Earth can be observed, described, and predicted
Grades 3–5
ESS1.B Earth and the Solar System. The Earth’s orbit and rotation, and the orbit of the moon around the Earth cause observable patterns.
ESS2.D Weather & Climate. Climate describes patterns of typical weather conditions over different scales and variations. Historical weather patterns can be analyzed so that they can make predictions about what kind of weather might happen next.
Grades 6–8
ESS1.B Earth and the Solar System. The solar system contains many varied objects held together by gravity. Solar system models explain and predict eclipses, tides, lunar phases, and seasons.
ESS2.D Weather & Climate. Complex interactions determine local weather patterns and influence climate, including the role of the ocean.
PS1.A Structure of Matter. The fact that matter is composed of atoms and molecules can be used to explain the properties of substances, diversity of materials, states of matter, phase changes, and conservation of matter.
PS1.B Chemical Reactions. Reacting substances rearrange to form different molecules, but the number of atoms is conserved. Some reactions release energy and others absorb energy.
PS1.C Nuclear Processes. Nuclear fusion can result in the merging of two nuclei to form a larger one, along with the release of significantly more energy per atom than any chemical process. Nuclear fusion taking place in the cores of stars provides the energy released (as light) from those stars and produced all of the more massive atoms from primordial hydrogen.
PS4.A Wave Properties. A simple wave model has a repeating pattern with a specific wavelength, frequency, and amplitude, and mechanical waves need a medium through which they are transmitted. This model can explain many phenomena including sound and light. Waves can transmit energy
PS4.B Electromagnetic Radiation. The construct of a wave is used to model how light interacts with objects.
Grades 9–12
ESS1.B Earth and the Solar System. Kepler’s laws describe common features of the motions of orbiting objects. Observations from astronomy and space probes provide evidence for explanations of solar system formation. Changes in Earth’s tilt and orbit cause climate changes such as Ice Ages
ESS2.D Weather & Climate. The role of radiation from the sun and its interactions with the atmosphere, ocean, and land are the foundation for the global climate system. Global climate models are used to predict future changes, including changes influenced by human behavior and natural factors
PS1.A Structure of Matter. The sub-atomic structural model and interactions between electric charges at the atomic scale can be used to explain the structure and interactions of matter, including chemical reactions and nuclear processes. Repeating patterns of the periodic table reflect patterns of outer electrons. A stable molecule has less energy than the same set of atoms separated; one must provide at least this energy to take the molecule apart
PS1.B Chemical Reactions. Chemical processes are understood in terms of collisions of molecules, rearrangement of atoms, and changes in energy as determined by properties of elements involved.
PS1.C Nuclear Processes. Nuclear processes, including fusion, fission, and radio-active decays of unstable nuclei, involve changes in nuclear binding energies. The total number of neutrons plus protons does not change in any nuclear process. Strong and weak nuclear interactions determine nuclear stability and processes. Normal stars cease producing light after having converted all of the material in their cores to carbon or, for more massive stars, to iron. Elements more massive than iron are formed by fusion processes but only in the extreme conditions of supernova explosions, which explains why they are relatively rare.
PS4.A Wave Properties. The wavelength and frequency of a wave are related to one another by the speed of the wave, which depends on the type of wave and the medium through which it is passing. Waves can be used to transmit information and energy.
PS4.B Electromagnetic Radiation. Both an electromagnetic wave model and a photon model explain features of electromagnetic radiation broadly and describe common applications of electromagnetic radiation.