Aerosols: FIM Chem Model
Details
Permalink to Details- Added to the Catalog
- Available for
- SOS
- Explorer
- Categories
- Air: Chemistry
- Keywords
- Aerosols
- Anthropogenic Emissions
- Atmosphere
- Black Carbon
- Chemistry
- Climate
- Dust
- Emissions
- FIM
- Industrial
- Model
- Organic Carbon
- Soot
Description
Permalink to DescriptionThe Flow Following Finite Volume Icosahedral Model (FIM) was developed by NOAA to produce weather forecasts. In fact, weather forecasts from the FIM model are available for SOS here. Building upon the success of the FIM model, the FIM-Chem model was created. The FIM-Chem is the FIM model with chemistry and aerosol modules added. Aerosols are one of the biggest uncertainties in climate models due to their varied affects on radiation and cloud physics. The FIM-Chem allows researchers to forecast and study the behavior of aerosols in the atmosphere, leading to the potential for better Earth system modeling for climate prediction.
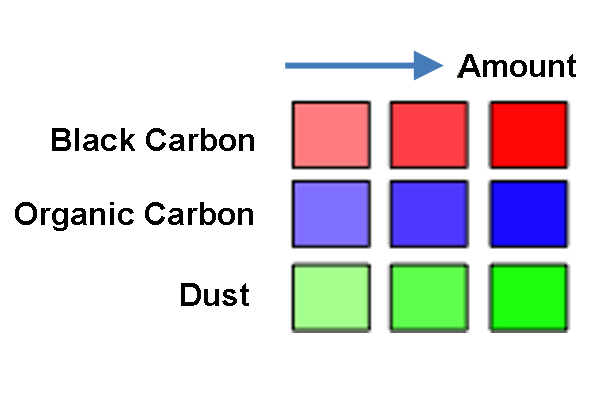
In this dataset, the FIM-Chem model is used to trace the presence of three aerosols from August 27, 2009 through September 7, 2009. Green shading is dust, red shading is black carbon aerosols and blue is organic carbon aerosols. When there are multiple aerosols present in one location, the color for the aerosols is combined. The two types of carbon often occur together, though in different proportions. Areas of anthropogenic emissions tend to have a redder color while areas of burning, such as wildfires, tend to have a more purplish-pink color because the ratio of organic carbons to black carbons varies for the two emissions sources. Wildfires have a high ratio of organic carbon to black carbon.
During the time period of this dataset there were wildfires in California, Alaska and British Columbia and biomass burning in Sub-Equatorial Africa and the Amazon region. The white shading is from the presence of all three aerosols. This can be seen in southern Europe where the anthropogenic emissions from Europe are mixing with the dry, dusty air in the Sahara.
Next Generation Science Standards
Permalink to Next Generation Science StandardsCross-cutting Concepts
Permalink to Cross-cutting ConceptsGrades 3–5
C2 Cause and Effect. Students routinely identify and test causal relationships and use these relationships to explain change. They understand events that occur together with regularity might or might not signify a cause and effect relationship
C3 Scale Proportion and Quantity. Students recognize natural objects and observable phenomena exist from the very small to the immensely large. They use standard units to measure and describe physical quantities such as weight, time, temperature, and volume.
C4 Systems and System Models. Students understand that a system is a group of related parts that make up a whole and can carry out functions its individual parts cannot. They can also describe a system in terms of its components and their interactions.
C7 Stability and Change. Students measure change in terms of differences over time, and observe that change may occur at different rates. Students learn some systems appear stable, but over long periods of time they will eventually change.
Grades 6–8
C2 Cause and Effect. Students classify relationships as causal or correlational, and recognize that correlation does not necessarily imply causation. They use cause and effect relationships to predict phenomena in natural or designed systems. They also understand that phenomena may have more than one cause, and some cause and effect relationships in systems can only be described using probability.
C3 Scale Proportion and Quantity. Students observe time, space, and energy phenomena at various scales using models to study systems that are too large or too small. They understand phenomena observed at one scale may not be observable at another scale, and the function of natural and designed systems may change with scale. They use proportional relationships (e.g., speed as the ratio of distance traveled to time taken) to gather information about the magnitude of properties and processes. They represent scientific relationships through the use of algebraic expressions and equations
C4 Systems and System Models. Students can understand that systems may interact with other systems; they may have sub-systems and be a part of larger complex systems. They can use models to represent systems and their interactions—such as inputs, processes and outputs—and energy, matter, and information flows within systems. They can also learn that models are limited in that they only represent certain aspects of the system under study.
C7 Stability and Change. Students explain stability and change in natural or designed systems by examining changes over time, and considering forces at different scales, including the atomic scale. Students learn changes in one part of a system might cause large changes in another part, systems in dynamic equilibrium are stable due to a balance of feedback mechanisms, and stability might be disturbed by either sudden events or gradual changes that accumulate over time
Grades 9–12
C2 Cause and Effect. Students understand that empirical evidence is required to differentiate between cause and correlation and to make claims about specific causes and effects. They suggest cause and effect relationships to explain and predict behaviors in complex natural and designed systems. They also propose causal relationships by examining what is known about smaller scale mechanisms within the system. They recognize changes in systems may have various causes that may not have equal effects.
C3 Scale Proportion and Quantity. Students understand the significance of a phenomenon is dependent on the scale, proportion, and quantity at which it occurs. They recognize patterns observable at one scale may not be observable or exist at other scales, and some systems can only be studied indirectly as they are too small, too large, too fast, or too slow to observe directly. Students use orders of magnitude to understand how a model at one scale relates to a model at another scale. They use algebraic thinking to examine scientific data and predict the effect of a change in one variable on another (e.g., linear growth vs. exponential growth).
C4 Systems and System Models. Students can investigate or analyze a system by defining its boundaries and initial conditions, as well as its inputs and outputs. They can use models (e.g., physical, mathematical, computer models) to simulate the flow of energy, matter, and interactions within and between systems at different scales. They can also use models and simulations to predict the behavior of a system, and recognize that these predictions have limited precision and reliability due to the assumptions and approximations inherent in the models. They can also design systems to do specific tasks.
C7 Stability and Change. Students understand much of science deals with constructing explanations of how things change and how they remain stable. They quantify and model changes in systems over very short or very long periods of time. They see some changes are irreversible, and negative feedback can stabilize a system, while positive feedback can destabilize it. They recognize systems can be designed for greater or lesser stability
Disciplinary Core Ideas
Permalink to Disciplinary Core IdeasGrades 3–5
ESS2.D Weather & Climate. Climate describes patterns of typical weather conditions over different scales and variations. Historical weather patterns can be analyzed so that they can make predictions about what kind of weather might happen next.
ESS3.A Natural Resources. Energy and fuels humans use are derived from natural sources and their use affects the environment. Some resources are renewable over time, others are not.
ESS3.C Human Impact on Earth systems. Societal activities have had major effects on the land, ocean, atmosphere, and even outer space. Societal activities can also help protect Earth’s resources and environments.
Grades 6–8
ESS2.D Weather & Climate. Complex interactions determine local weather patterns and influence climate, including the role of the ocean.
ESS3.A Natural Resources. Humans depend on Earth’s land, ocean, atmosphere, and biosphere for different resources, many of which are limited or not renewable. Resources are distributed unevenly around the planet as a result of past geologic processes
ESS3.B Natural Hazards. Mapping the history of natural hazards in a region and understanding related geological forces can help forecast the locations and likelihoods of future events, such as volcanic eruptions, earthquakes and severe weather.
ESS3.C Human Impact on Earth systems. Human activities have altered the biosphere, sometimes damaging it, although changes to environments can have different impacts for different living things. Activities and technologies can be engineered to reduce people’s impacts on Earth.
ESS3.D Global Climate Change. Human activities affect global warming. Decisions to reduce the impact of global warming depend on understanding climate science, engineering capabilities, and social dynamics.
Grades 9–12
ESS2.D Weather & Climate. The role of radiation from the sun and its interactions with the atmosphere, ocean, and land are the foundation for the global climate system. Global climate models are used to predict future changes, including changes influenced by human behavior and natural factors
ESS3.A Natural Resources. Resource availability has guided the development of human society and use of natural resources has associated costs, risks, and benefits.
ESS3.B Natural Hazards. Natural hazards and other geological events have shaped the course of human history at local, regional, and global scales. Human activities can contribute to the frequency and intensity of some natural hazards.
ESS3.C Human Impact on Earth systems. Sustainability of human societies and the biodiversity that supports them requires responsible management of natural resources, including the development of technologies that produce less pollution and waste and that preclude ecosystem degradation.
ESS3.D Global Climate Change. Global climate models used to predict changes continue to be improved, although discoveries about the global climate system are ongoing and continually needed.
PS1.B Chemical Reactions. Chemical processes are understood in terms of collisions of molecules, rearrangement of atoms, and changes in energy as determined by properties of elements involved.
PS4.B Electromagnetic Radiation. Both an electromagnetic wave model and a photon model explain features of electromagnetic radiation broadly and describe common applications of electromagnetic radiation.
Notable Features
Permalink to Notable Features- Areas of anthropogenic emissions tend to have a redder color while areas of burning, such as wildfires, tend to have a more purplish-pink color
- Areas where all three aerosols are present have a white color.
- During the time period of this dataset there were wildfires in California, Alaska and British Columbia and biomass burning in Sub-Equatorial Africa and the Amazon region